| Metric | Air Drying | Genevac EZ-2 |
|---|---|---|
| Assay 1 — “no calls” (no usable data) | 59.64% | 1.82% |
| Assay 2 — “no calls” (no usable data) | 45.57% | 11.72% |
| Contamination Risk | Higher (multi-day open exposure) | Minimal (sealed, aseptic) |
| Time to Concentrate | Multiple days | Under 1 hour |
The “Basic Principles of Freeze-Drying” is a six-part series that presents the fundamental principles and practical considerations of freeze-drying (lyophilization). The topics are arranged to follow the sequence of a typical freeze-drying process, beginning with the basic physical principles of sublimation and low temperature drying, followed by the equipment used to create the required processing environment.
Subsequent articles examine product containers, containment systems, and material properties that influence freezing behavior, drying performance, and final product characteristics. These factors are then applied to the design of a freeze-drying cycle, including freezing, primary drying, and secondary drying.
The final sections address cycle optimization, scale-up to production-sized equipment, and long-term product stability. Together, these articles provide a structured overview of the variables that must be understood and controlled in order to develop, operate, and maintain a successful freeze-drying process.
Introduction
Freeze-drying is the removal of ice or other frozen solvents from a material through the process of sublimation and the removal of bound water molecules through the process of desorption.
Lyophilization and freeze-drying are terms that are used interchangeably depending on the industry and location where the drying is taking place. Controlled freeze-drying keeps the product temperature low enough during the process to avoid changes in the dried product appearance and characteristics. It is an excellent method for preserving a wide variety of heat-sensitive materials such as proteins, microbes, pharmaceuticals, tissues & plasma.
How Sublimation Makes Freeze-Drying Possible
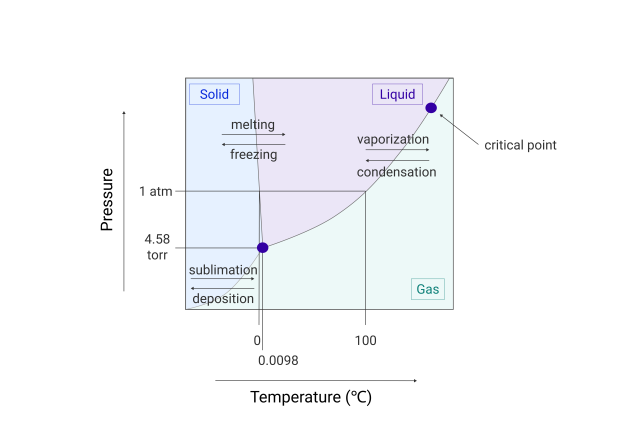
Sublimation is when a solid (ice) changes directly to a vapor without first going through a liquid (water) phase. Thoroughly understanding the concept of sublimation is a key building block to gaining knowledge of freeze-drying.
Sublimation is a phase change and heat energy must be added to the frozen product for it to occur. Sublimation in the freeze-drying process can be described simply as:
- FREEZE – The product is completely frozen, usually in a vial, flask or tray
- VACUUM – The product is then placed under a deep vacuum, well below the triple point of water
- DRY – Heat energy is then added to the product causing the ice to sublime
Lyophilization Step-by-Step
The steps required to lyophilize a product in a batch process can be summarized as follows:
- Pretreatment / formulation
- Loading / container (bulk, flask, vials)
- Freezing (thermal treatment) at atmospheric pressure
- Primary Drying (sublimation) under vacuum
- Secondary Drying (desorption) under vacuum
- Backfill & Stoppering (for product in vials) under partial vacuum
- Removal of dried product from freeze dryer
In addition to providing an extended shelf life, successful freeze-drying should yield a product that has a short reconstitution time with acceptable potency levels. The process should be repeatable with well-defined temperature, pressure, and time parameters for each step. Visual and functional characteristics of the dried product are also important for many applications.

These fundamental steps — freezing, vacuum, and controlled heat input — form the basis of every freeze-drying process. In the next article, these principles will be applied to the freeze dryer itself, where refrigeration, vacuum, and vapor capture systems create the controlled environment required for sublimation.









